Antonio Martinez Sánchez
Professor in Computer Sciences, University of Murcia, Spain
Date and time: June 5th, 2024 Wednesday, at 8am PDT / 11am EDT / 3pm BST / 5pm CEST / 11pm China
For the zoom links, please join the One World Cryo-EM mailing list.
Fast Normalized Cross-Correlation for Template Matching with Rotations
Object detection is a main task in computer vision. Template matching is the reference method for detecting objects with arbitrary templates. However, template matching computational complexity depends on the rotation accuracy, being a limiting factor for large 3D images (tomograms).
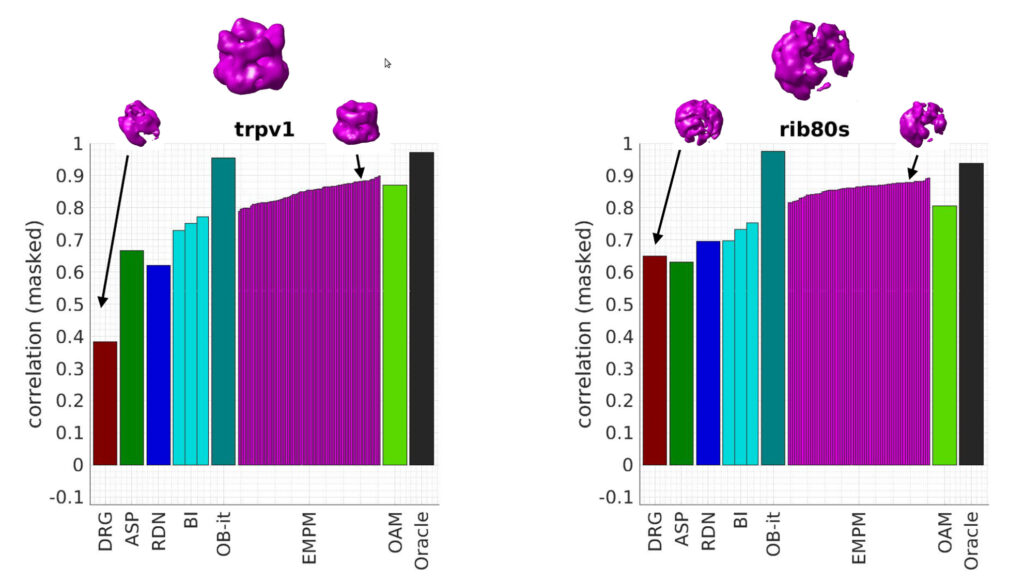
Here, we implement a new algorithm called tensorial template matching, based on a mathematical framework that represents all rotations of a template with a tensor field. Contrary to standard template matching, the computational complexity of the presented algorithm is independent of the rotation accuracy.
Using both, synthetic and real data from cryo-electron tomography, we demonstrate that tensorial template matching is much faster than template matching and has the potential to improve its accuracy.